science news for your neurons
Chemistry Labs
Download the Rubric for online lab experiments below.
| lab_report_chemistry.docx | |
| File Size: | 15 kb |
| File Type: | docx |
Available Labs
1-Measurement, 2-Scientific Method, 3-Organic Molecules, 4-Cells & Membranes, 5-Respiration, 6-Cell Cycle
7-Genetics, 8-DNA, 9-Electrophoresis, 10-Plants, 11-Evolution, 12-Animals
Click the button below to begin an online lab.
1-Measurement, 2-Scientific Method, 3-Organic Molecules, 4-Cells & Membranes, 5-Respiration, 6-Cell Cycle
7-Genetics, 8-DNA, 9-Electrophoresis, 10-Plants, 11-Evolution, 12-Animals
Click the button below to begin an online lab.
Lesson 1 boyle's law

Before we start, who is Robert Boyle? Click the link to find out more about Robert Boyle. http://chemistry.about.com/od/famouschemists/p/boylebio.htm
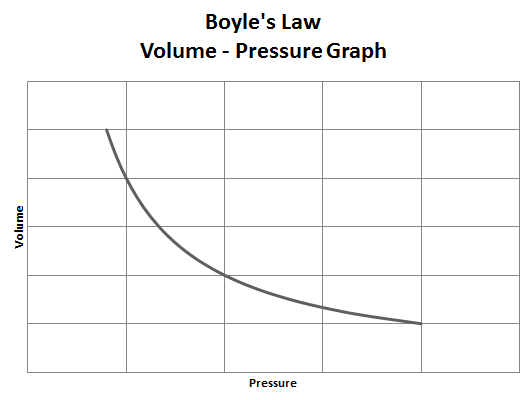
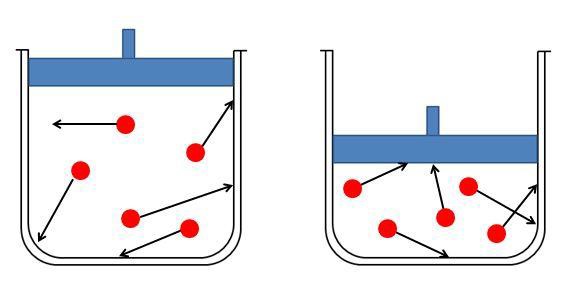
Now that we know a little about Mr. Boyle, lets take a look at the law that he came up with. Boyle's Law states that the volume of a given amount of gas held at a constant temperature varies inversely with the pressure. The relationship between pressure and volume of Boyle's Law is expressed in mathematical terms as P1V1=P2V2.
Our friends from brightstorm have a great presentation, check it out by clicking play below.
Now that we know a little about Mr. Boyle, lets take a look at the law that he came up with. Boyle's Law states that the volume of a given amount of gas held at a constant temperature varies inversely with the pressure. The relationship between pressure and volume of Boyle's Law is expressed in mathematical terms as P1V1=P2V2.
Our friends from brightstorm have a great presentation, check it out by clicking play below.
So once again Boyle’s Law states the relationship between pressure and volume. This is an inverse relationship which means that as the volume of a gas is decreased, the pressure exerted by the gas increases. This relationship can be represented in a mathematical equation.
Formula:
Equation 1 can be rearranged to make Equation 2
Equation 1 Equation 2
V 1P 1 = V 2 P 2 V 1/V2 = P 2/P 1
Example Problem
A sample of a gas occupies a volume of 120.4 mL in a balloon at a pressure of 1.24 atm. Determine the volume of the balloon when the pressure outside the balloon is decreased to 1.14 atm.
Variable Assignment
V1 = 120.4 mL
P1 = 1.24 atm
V2 = X (solve for this variable)
P2 = 1.14 atm
Step One: Separate the variable
Start with standard equation V 1P 1 = V 2 P 2
Divide both sides by P2 V 1P 1/P2 = V 2 P 2/ P 2
Set equation equal to V2 V 1P 1/P2 = V 2
Step Two: Substitute in for the variables
(120.4mL)(1.24atm)/(1.14atm)= V 2
Step Three: Solve for the indicated variable
V2 = 131 mL
Now you can give it a try!
WORKSHEET
1. A container has a volume of 5.60 liters, with a pressure of 1.25 atm. What would be the new pressure if the volume was decreased to 0.950 liters? [Difficulty 1]
2. A balloon with a volume of 250.0 ml at sea level has a pressure of 760.0 mm of Hg. The balloon is released and it climbs to a level with a new pressure of 600.0 mm of Hg. What is the new volume of the balloon? [Difficulty 1]
3. A healthy male adult has a lung capacity around 6.00 liters. The pressure in the lungs at that volume is measured to be 755 mm of Hg. If the lungs are compressed to a new volume of 3.81 liters, what would be the new pressure in the lungs? What would happen to the air in the lungs? [Difficulty 2]
4. One of the pistons in your car engine has a capacity of around 930.0 ml at normal atmospheric pressure of 765.0 mm of Hg. If a piston compresses the air in the cylinder to 20 % of its original volume, what would be the pressure of the gas? [Difficulty 2]
5. A container has a total volume of 1.250 L. Of this total volume, there is 125.0 mL of headspace (the area above the liquid) for CO2 gas put in to carbonate the liquid. The pressure of this gas is 1.58 atm. The valve at the bottom of the container is opened to allow liquid to be removed until the volume of the liquid is 0.750 L. What is the resulting pressure of the gas above the liquid? [Difficulty 2]
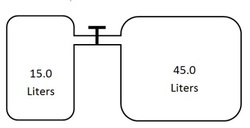
6. Two containers are attached together as shown with a valve between the containers.
The 15.0 L flask contains gas at a pressure of 3.15 atmospheres. The 45.0 L container does not contain any gas. The value between the two containers is opened and the gas in the 15.0 L container then also fills the 45.0 L container. What is the resulting pressure of the gas in the two containers? [Difficulty 3]
The 15.0 L flask contains gas at a pressure of 3.15 atmospheres. The 45.0 L container does not contain any gas. The value between the two containers is opened and the gas in the 15.0 L container then also fills the 45.0 L container. What is the resulting pressure of the gas in the two containers? [Difficulty 3]
7. Air pressure is measured in different units. Two common units are PSI (pounds per square inch) and millimeters of mercury (mm Hg) which is commonly used in a barometer. The highest barometer reading ever recorded is just over 15.6 PSI. The lowest barometer reading ever recorded was in the middle of about 12.5 PSI. These are all sea level readings. In Denver the average pressure is 12.1 PSI. What would the pressure be in mm of Hg? If you had a beach ball at the beach with a volume of 5.65 liters where the pressure is 750.0 mm of Hg, what would be the volume of the ball when you flew to Denver? What would happen to the ball? [Difficulty 3]
Here is a great link to videos for 1st year chemistry students. https://www.khanacademy.org/science/chemistry